When you mix things in a chemical reaction, not all parts get used up the same way. One part often runs out first and stops the reaction. This is called the limiting reactant. In this article, we will look at how to express limiting reactant in chemical formula. We start with the basics and go step by step. You will see why it matters and how to find it in real examples. This helps in school labs or even in big factories where chemicals are made.
The idea comes from stoichiometry, which is like a recipe for chemicals. Just as in baking, if you have too little flour, you can’t make all the cookies you want. The same happens in reactions. We use balanced equations to show the right amounts. But when amounts don’t match, we need to find the limiting one. Let’s dive in and make it clear.
What Is a Limiting Reactant?
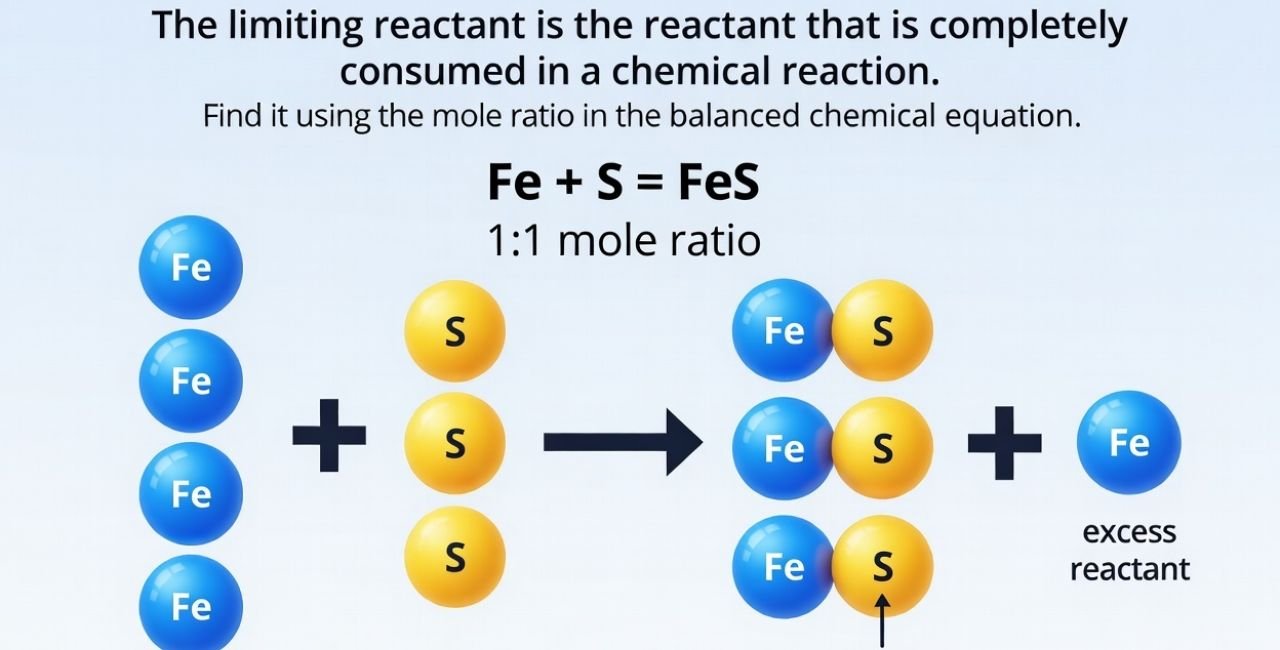
A limiting reactant is the substance in a chemical reaction that gets used up first. It limits how much product you can make. The other substances are called excess reactants because some of them are left over.
Think of it like making sandwiches. If you have 4 slices of bread and 3 pieces of cheese, you can only make 1 sandwich if each needs 2 bread and 1 cheese. The cheese limits you, even if you have extra bread.
In chemistry, this happens all the time. Reactions follow a balanced chemical equation, which shows the ratio of reactants. If the amounts you start with don’t match that ratio, one will limit the others.
Why Does It Matter?
Knowing the limiting reactant helps predict how much product forms. This is key in labs to avoid waste. In industries, it saves money by using just the right amounts. It also helps understand reaction efficiency.
For example, in making medicine or fuels, getting this right means better results and less leftover stuff.
Background of the Concept
The idea of limiting reactants ties to stoichiometry, which Antoine Lavoisier helped develop in the 1700s. He showed that mass is conserved in reactions. Later, in the 1800s, chemists like John Dalton built on atomic theory, leading to balanced equations.
By the 1900s, this became standard in teaching chemistry. Textbooks started including examples to show how amounts affect outcomes. Today, it’s a core part of high school and college courses.
Achievements in this area include better chemical engineering. For instance, in World War II, it helped optimize explosive production without waste.
How to Identify the Limiting Reactant
To find the limiting reactant, follow these steps. We use moles because equations are based on molecule ratios, not grams.
Bold Steps for Clarity:
- Balance the Equation: Make sure the chemical formula shows equal atoms on both sides.
- Convert Masses to Moles: Use molar mass. Moles = mass / molar mass.
- Compare Ratios: See the equation’s coefficients. Calculate how much of one reactant is needed for the other.
- Find the One That Runs Out First: The reactant that requires more than what’s available is limiting.
Let’s use an example from a trusted source. In the reaction N₂ + 3H₂ → 2NH₃, suppose you have 28 grams of N₂ and 6 grams of H₂.
- Molar mass N₂ = 28 g/mol, so 28/28 = 1 mole.
- Molar mass H₂ = 2 g/mol, so 6/2 = 3 moles.
The equation says 1 mole N₂ needs 3 moles H₂. Here, you have exactly that, so neither limits; both are used up.
But if you have 28 grams N₂ (1 mole) and only 4 grams H₂ (2 moles), then H₂ limits because you need 3 moles but have only 2.
You can learn more about this reaction on Brainly.
How to Express Limiting Reactant in Chemical Formula
This section focuses on how to express limiting reactant in chemical formula. It’s not about changing the formula but showing it in calculations.
In a balanced equation like 2Mg + O₂ → 2MgO, the formula is fixed. To express the limiting one, we note the amounts.
For instance:
- Write the equation: 2Mg + O₂ → 2MgO
- State amounts: 2.4 grams Mg, 10 grams O₂.
- Calculate moles: Mg = 2.4/24 = 0.1 moles, O₂ = 10/32 = 0.3125 moles.
- Ratio: 2 moles Mg per 1 mole O₂.
- Needed O₂ for Mg: 0.1 / 2 = 0.05 moles O₂.
- Since 0.3125 > 0.05, Mg limits.
We express it by saying “Mg is the limiting reactant in the formula 2Mg + O₂ → 2MgO with given amounts.”
Sometimes, we denote it in stoichiometry problems as LR for limiting reactant next to the symbol.
Steps to Calculate Limiting Reactant
Here is a detailed guide. We break it into simple parts.
Step 1: Balance the Equation
Always start here. For example, magnesium and oxygen: 2Mg + O₂ → 2MgO.
This shows 2 atoms Mg react with 1 molecule O₂.
Step 2: Find Moles of Each Reactant
Use periodic table for molar masses.
- Mg: 24 g/mol
- O: 16 g/mol, so O₂ = 32 g/mol
If you have 4.8 grams Mg: 4.8 / 24 = 0.2 moles.
10 grams O₂: 10 / 32 ≈ 0.31 moles.
Step 3: Use Stoichiometric Ratios
From equation, moles Mg / 2 = moles O₂ / 1
So, for Mg: 0.2 / 2 = 0.1 moles O₂ needed.
You have 0.31, which is more, so Mg limits.
You can also find product from each.
From Mg: 0.2 moles Mg × (2 moles MgO / 2 moles Mg) = 0.2 moles MgO.
From O₂: 0.31 moles O₂ × (2 moles MgO / 1 mole O₂) = 0.62 moles MgO.
Least product is from Mg, so it limits.
Step 4: Calculate Excess
Excess O₂ = initial – used.
Used O₂ = 0.2 moles Mg × (1 mole O₂ / 2 moles Mg) = 0.1 moles.
Excess = 0.31 – 0.1 = 0.21 moles.
Examples of Limiting Reactants
Let’s look at several cases to make it clear.
Example 1: Ammonia Synthesis
N₂ + 3H₂ → 2NH₃
With 14 grams N₂ (0.5 moles) and 6 grams H₂ (3 moles).
Needed H₂ for N₂: 0.5 × 3 = 1.5 moles.
Have 3 > 1.5, so N₂ limits.
Product: 0.5 × 2 = 1 mole NH₃.
Excess H₂: 3 – 1.5 = 1.5 moles.
This is a common industrial reaction, called Haber process.
Example 2: Glucose Respiration
C₆H₁₂O₆ + 6O₂ → 6CO₂ + 6H₂O
25 grams glucose (180 g/mol) = 0.139 moles.
40 grams O₂ (32 g/mol) = 1.25 moles.
Needed O₂: 0.139 × 6 ≈ 0.833 moles.
Have 1.25 > 0.833, glucose limits.
Product CO₂: 0.139 × 6 ≈ 0.833 moles.
This shows in biology how cells use sugar.
From LibreTexts.
Example 3: Magnesium Oxide
2Mg + O₂ → 2MgO
2.4 grams Mg = 0.1 moles.
3.2 grams O₂ = 0.1 moles.
Ratio 2:1, so for 0.1 moles Mg, need 0.05 moles O₂.
Have 0.1 > 0.05, Mg limits? Wait, check.
Moles Mg / coeff = 0.1 / 2 = 0.05
Moles O₂ / 1 = 0.1
Smaller is Mg, yes.
Example 4: Complex One
4C₂H₃Br₃ + 11O₂ → 8CO₂ + 6H₂O + 6Br₂
76.4 grams C₂H₃Br₃ (molar 266.7) ≈ 0.286 moles.
49.1 grams O₂ ≈ 1.53 moles.
Needed O₂: 0.286 × (11/4) ≈ 0.786 moles.
Have 1.53 > 0.786, C₂H₃Br₃ limits.
Example 5: Sodium Peroxide
2Na₂O₂ + 2H₂O → 4NaOH + O₂
78 grams Na₂O₂ (78 g/mol) = 1 mole.
18 grams H₂O = 1 mole.
Ratio 2:2 or 1:1.
Equal, no limiting.
But if 78 grams Na₂O₂ and 29.4 grams H₂O (1.63 moles), Na₂O₂ limits? Molar Na₂O₂ 78, yes 1 mole, H₂O 18 g/mol, 29.4/18=1.63.
Needed H₂O: 1 × (2/2)=1 mole.
Have 1.63 >1, Na₂O₂ limits.
More Examples
- CoO and O₂: 4CoO + O₂ → 2Co₂O₃
24.5 grams CoO (75 g/mol)≈0.327 moles.
2.58 grams O₂≈0.081 moles.
Ratio 4:1, needed O₂ for CoO: 0.327/4≈0.082.
Have 0.081 <0.082, O₂ limits.
- SiO₂ + 2H₂F₂ → SiF₄ + 2H₂O
28.7 grams SiO₂ (60 g/mol)≈0.478 moles.
22.6 grams H₂F₂ (assume HF, but H₂F₂ is 38 g/mol?) Wait, probably H₂F₂ is hypothetical, but calculation similar.
These show variety.
Real-World Applications
In medicine, limiting reactants ensure right doses in drug synthesis.
In environment, understanding limits helps in pollution control, like in car exhaust.
Statistics: In chemical industry, optimizing limits can reduce waste by 20-30%, per reports.
In food, fermentation uses sugar as limiting to control alcohol.
For more on chemistry in daily life, check NY Today Magazine.
Percent Yield and Related Concepts
Once you find limiting, calculate theoretical yield: max product from limiting.
Actual yield is what you get in lab.
Percent yield = (actual / theoretical) × 100.
Why less? Side reactions, impurities.
Example: In MgO, theoretical 4 grams, actual 3.5, yield 87.5%.
From Khan Academy.
Tips for Students
- Always check units: grams to moles.
- Practice with simple equations first.
- Use tables for organization:
| Reactant | Mass | Molar Mass | Moles | Coefficient | Moles / Coeff |
|———-|——|————|——-|————-|—————|
| Mg | 2.4 | 24 | 0.1 | 2 | 0.05 |
| O₂ | 10 | 32 | 0.312| 1 | 0.312 |
Smallest moles/coeff is limiting.
- Don’t forget to balance!
Common Mistakes
- Forgetting to convert to moles.
- Mixing up limiting and excess.
- Not using balanced equation.
To avoid, double-check calculations.
Advanced Topics
In multi-step reactions, limiting carries over.
In gases, use volumes if at same temp/pressure.
In solutions, concentrations matter: moles = concentration × volume.
Example: 0.1 M NaOH 100 ml = 0.01 moles.
FAQs
What is how to express limiting reactant in chemical formula?
It’s showing which reactant limits in the balanced equation by calculating amounts.
How do I find it without masses?
If ratios match, none; else, compare.
Can there be more than one limiting?
Rare, usually one.
Why use moles not grams?
Because equations are by particles, not weight.
Conclusion
We covered how to express limiting reactant in chemical formula from basics to examples. Remember, it’s about finding which runs out first using stoichiometry. This helps in predictions and efficiency.
In summary, balance the equation, convert to moles, compare ratios, and calculate. With practice, it becomes easy.
What chemical reaction have you tried where you think there was a limiting reactant? Share in comments!
References
- LibreTexts – Comprehensive guide with examples and steps. Great for students needing detailed calculations.
- Brainly – Community answers on specific reactions like ammonia. Helps with quick problem-solving.
- Khan Academy – Explains yields and limits with videos. Ideal for visual learners.
